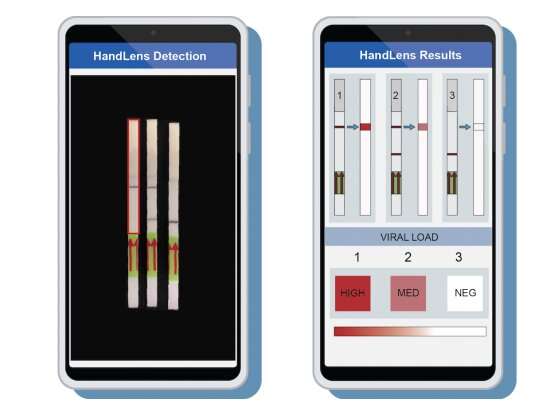
One of the keys to bringing a viral outbreak under control is rapid detection and diagnosis, which depend on the availability of fast, low-cost, easy-to-use tests that don’t require labs or expensive equipment to process them. Scientists at the Broad Institute of MIT and Harvard and collaborators in the United States, Nigeria, and Sierra Leone have now validated such tests for Ebola and Lassa—two of the deadliest and most transmissible human viruses—in settings with limited infrastructure. The work appears in Nature Communications.
The diagnostic tests use the CRISPR-based SHERLOCK assay to detect low levels of virus in patient samples and generate either a fluorescent readout or a result on a paper strip. The test can be tailored to detect specific viruses from certain regions, requires only a simple heat block and basic supplies to run, costs less than US$1 per sample, could be used on saliva or urine—eliminating the need for blood draws—and can return results in less than an hour.
The tests also use a rapid chemical and heat treatment called HUDSON to inactive the virus in patient samples. HUDSON makes the patient samples safer for clinical staff to handle in a low-tech environment, and eliminates the need to extract a virus’s genetic material from the samples before analyzing.
The research team was led at Broad by Kayla Barnes, an NIH Fogarty K fellow at the Harvard School of Public Health and Broad Institute, Anna Lachenauer, a medical student at Stanford University School of Medicine, and institute member Pardis Sabeti, a professor at Harvard University and investigator with the Howard Hughes Medical Institute.
https://youtube.com/watch?v=9XI8jDszqWs%3Fcolor%3Dwhite
To showcase SHERLOCK’s field utility, team members led by Christian Happi at Redeemer’s University in Nigeria deployed a Lassa-specific assay during a recent Lassa fever outbreak—the first use of SHERLOCK in a low/middle income country. The team also compared the diagnostic against a standard RT-qPCR assay for Lassa.
Collaborators at Kenema Government Hospital in Sierra Leone and at the US Army Medical Research Institute of Infectious Diseases benchmarked an Ebola-specific version of the SHERLOCK assay, using samples collected during the 2014-16 outbreak in Sierra Leone and more recent outbreaks in the Democratic Republic of the Congo. The NIH Integrated Research Facility also validated HUDSON’s ability to heat-inactivate Ebola virus in their BL4 facilities, further establishing the safety and efficiency of this step.
The SHERLOCK assays performed as consistently as, or better than, other diagnostics in these validations—demonstrating the platform’s potential for clinical use in the future in resource-limited areas.
Source: Read Full Article